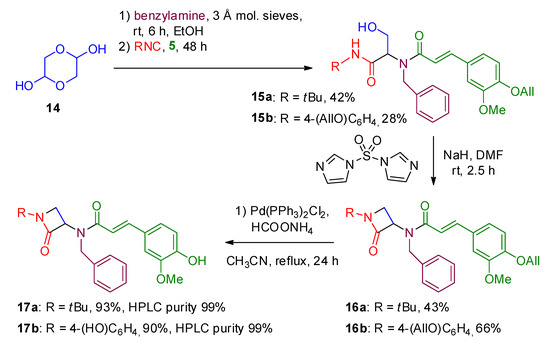
A role for ring‐closing metathesis in medicinal chemistry: Mimicking secondary architectures in bioactive peptides - Pérez de Vega - 2011 - Medicinal Research Reviews - Wiley Online Library

A role for ring‐closing metathesis in medicinal chemistry: Mimicking secondary architectures in bioactive peptides - Pérez de Vega - 2011 - Medicinal Research Reviews - Wiley Online Library

PDF) A new simple route to deoxyamino sugars from non-sugar material: synthesis of d-tolyposamine and 4-epi-d-tolyposamine and formal synthesis of d-vicenisamine | Yoshitaka Matsushima - Academia.edu

A role for ring‐closing metathesis in medicinal chemistry: Mimicking secondary architectures in bioactive peptides - Pérez de Vega - 2011 - Medicinal Research Reviews - Wiley Online Library

A role for ring‐closing metathesis in medicinal chemistry: Mimicking secondary architectures in bioactive peptides - Pérez de Vega - 2011 - Medicinal Research Reviews - Wiley Online Library
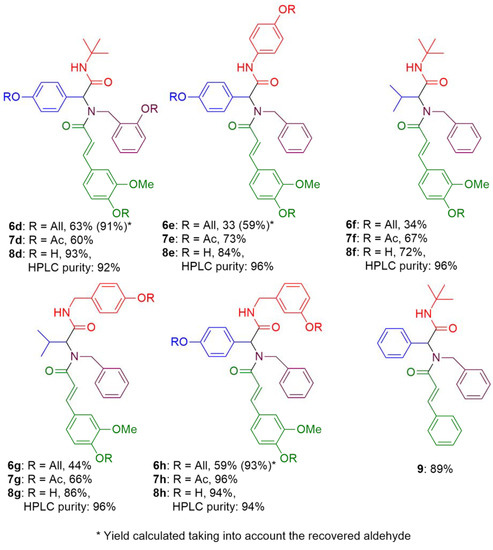
Molecules | Free Full-Text | Multicomponent Synthesis of Polyphenols and Their In Vitro Evaluation as Potential β-Amyloid Aggregation Inhibitors | HTML
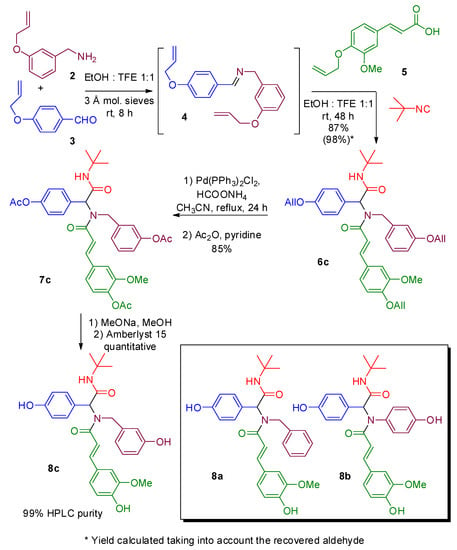
Molecules | Free Full-Text | Multicomponent Synthesis of Polyphenols and Their In Vitro Evaluation as Potential β-Amyloid Aggregation Inhibitors | HTML